Structure and function of lead-acid battery
- By: JinHan
- Jan 23,2023
follow us
The lead-acid battery is generally composed of 3 or 6 single cells in series, consisting of plates, separators, electrolyte, a shell, poles and a liquid filler plug (not available for maintenance free batteries).
1. Electrode plate of lead-acid battery
The electrode plate is divided into positive plate and negative plate, both of which are composed of grid frame and active substance.
Function of grid frame.
Adding ladder has side effects, which will accelerate the release of hydrogen and the consumption of electrolyte. It is also easy to be resolved from the positive plate grid frame, causing battery self discharge and grid frame corrosion, shortening the service life of the battery. At present, low brocade alloy grid frame (2% - 3.5% ladder content) or lead calcium alloy grid frame are mostly used at home and abroad.
The active substance on the positive plate is lead dioxide (Pb02), which is dark brown; The active substance on the negative plate is sponge like pure lead (Pb), which is green gray. The active material is mixed into paste to fill the gap of the grid frame and dried to form the electrode plate.
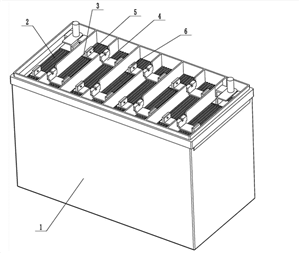
Immerse one positive and one negative plate into the electrolyte to obtain an electromotive force of about 2V. In order to increase the capacity of the battery, multiple positive and negative plates are often connected in parallel to form a positive and negative plate group, as shown in the figure (drawing). In each single cell, the number of positive plates is one less than that of negative plates, so that each positive plate is located between the two negative plates, which can make the discharge on both sides of the positive plate uniform, and avoid the arch of the plate due to uneven discharge.
2. Separator of lead-acid battery
The separator is inserted between the positive and negative plates to prevent short circuit caused by contact between the positive and negative plates. The separator shall be acid resistant and porous to facilitate electrolyte penetration. The commonly used partition materials are wood, microporous rubber and microporous plastic. Among them, the wooden partition has poor acid resistance and has been eliminated. The microporous rubber separator has the best performance but high cost. The microporous plastic separator has small aperture, high porosity and low cost, so it is widely used.
During installation, the side with groove should face the positive plate and be perpendicular to the bottom of the shell. At the same time, the groove can dredge the electrolyte up and down, so that bubbles can rise along the groove. The microporous plastic can be made into a bag separator, which can be sleeved on the positive plate to prevent active substances from falling off.
3. Electrolyte
In the chemical reaction of lead-acid battery, electrolyte plays the role of conducting electricity between ions and participates in the chemical reaction of battery. The electrolyte is made of pure sulfuric acid (H: S0.) and distilled water in a certain proportion, and its density is generally 1.24~1.30g/cm '. The electrolyte density is selected according to season and region. The liquid level shall be 10-15mm higher than the upper plane of the electrode plate (separator).
4. Shell of lead-acid battery
The shell is used for holding electrolyte and plate group, and should be acid resistant, heat resistant and shock resistant. The shell is mostly made of hard rubber or polypropylene plastic, which is an integral structure, and the bottom has raised ribs to place the plate group. The shell is divided into 3 or 6 disconnected cells by the partition wall, and each cell is connected in series with a lead mass link (as shown in the figure). The upper part of the shell is sealed with a battery cover made of the same material. The battery cover is provided with a liquid filling hole corresponding to each single cell cell for adding electrolyte and distilled water, and measuring the density, temperature and liquid level of electrolyte. The vent hole on the liquid filling hole cover can smoothly discharge the gas generated in the chemical reaction of the battery.
Connecting strip: the connecting strip is used to connect the single cell storage tanks in series, so as to improve the terminal voltage of the middle storage battery
5. Pole of lead-acid battery
The horizontal plate of the two pole plate group at the head and tail of the ordinary lead battery is welded with three types of terminals, namely, terminal side control type, cone type and I-type. In order to facilitate differentiation, the positive terminal is marked with "+" or "p", some terminals are painted with red paint, and the negative terminal is marked with "-" or "N". If the resolution is unclear, use a voltmeter to measure.
--End--

 English
English Russian
Russian Portuguese
Portuguese Arabic
Arabic Bangla
Bangla Indonesian
Indonesian